
When it comes to scuba diving, grasping the concept of neutral buoyancy is crucial. Yet, achieving neutral buoyancy is not a one-time accomplishment. It requires continuous adjustments to the air volume inside the buoyancy compensator (BC) based on the depth.
Why is it necessary to fine-tune the air volume within the BC as per the depth? As explained in our previous article, pressure undergoes changes as you descend deeper. Thus, we need to adjust the air inside the BC to alter its volume in response to pressure variations.
In this article, we will explore the correlation between pressure and volume. We will also delve into the reasons behind the need for these adjustments.
The Connection Between Pressure and Volume
Let’s take a trip down memory lane to when we were in elementary school during an excursion to a mountain. Remember those snacks we often couldn’t have because they were unhealthy? Picture this: after enjoying our lunch at the mountaintop, we reach into our backpacks and pull out a bag of potato chips. Surprisingly, you found it inflated!
The reason behind this unexpected occurrence is due to the increase in altitude. The decrease in atmospheric pressure leads the bag to expand. But why does this happen?
Like the relationship between water depth and pressure, pressure changes with altitude. The bag of chips expands because the air inside it undergoes expansion.
Gases consist of tiny molecules that move within a given space. After the bag of chips is sealed, it experiences pressure from the surrounding environment. Now, imagine what happen when the external pressure decreases. The molecules inside the bag become livelier, wouldn’t them?
When pressure decreases, the molecules inside the bag have more room to move, causing the volume to increase.
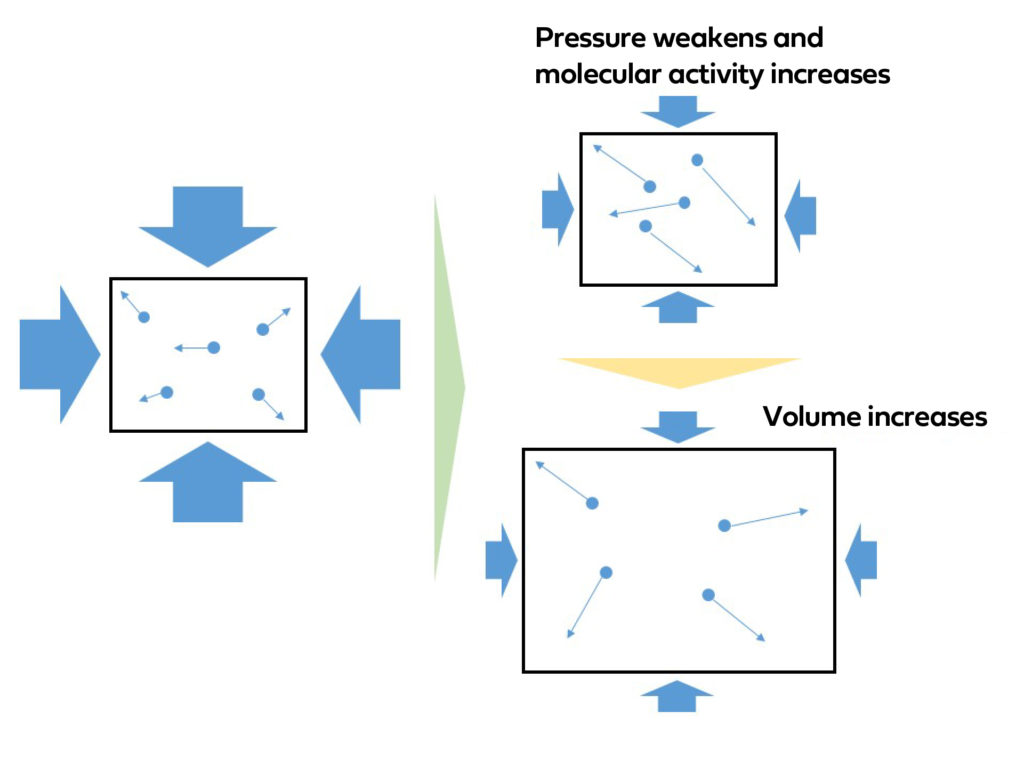
In essence, when pressure decreases, volume increases, and vice versa.
That’s right, volume is inversely proportional to pressure.
But what is the exact ratio of this inverse proportionality?
There is no need for any specific coefficient between pressure and volume. It’s simple as follow:
If pressure doubles, volume is halved.
If pressure triples, volume becomes one-third.
If pressure is halved, volume doubles.
If pressure is one-third, volume triples.
Easy to remember, isn’t it?
This connection between pressure and volume is explained by Boyles’ law.
The name may not seem important, but some educational institutions include questions about it in written exams. I would recommend remembering it!
Exploring the Connection with Neutral Buoyancy
Now, let’s delve into the focus of our discussion.
Achieving neutral buoyancy requires delicate adjustments to the amount of air within the BC (Buoyancy Compensator).
If you’ve been following along, you likely grasp this concept already!
The quantity of air inside the BC varies with changes in water depth.
For instance, at a depth of 30 meters, the pressure amounts to 4 atmospheres.
Let’s imagine that we established neutral buoyancy by adding air to the BC at this depth.
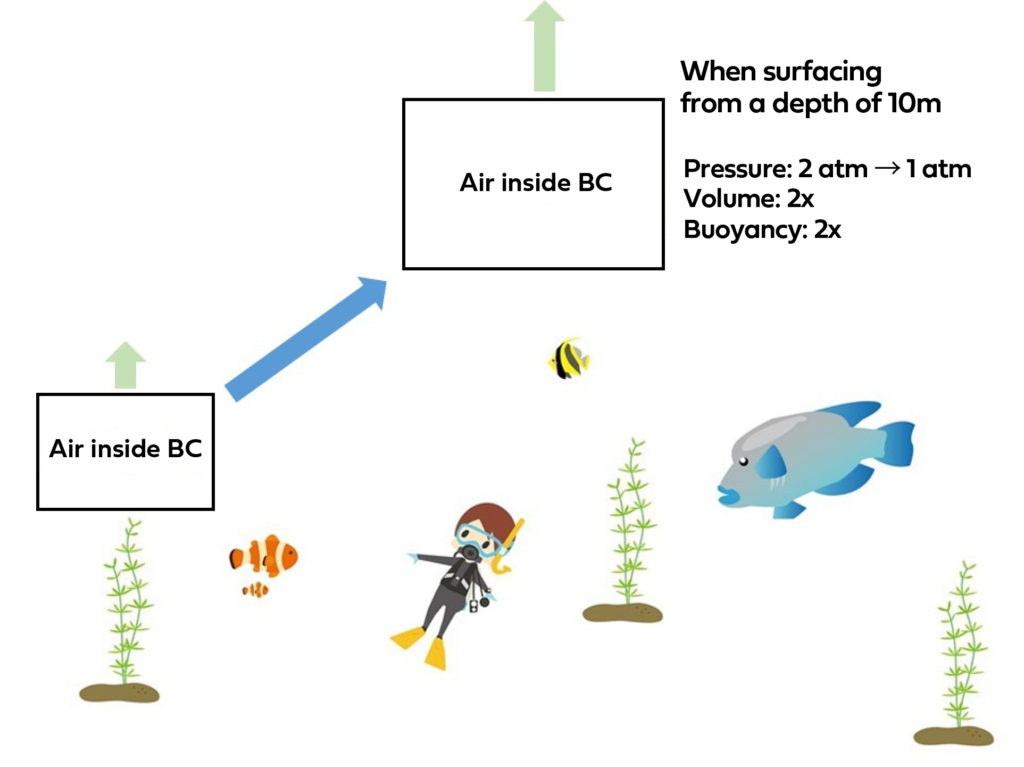
Now, let’s ascend to a shallower depth of 10 meters without manipulating the BC.
At this point, the pressure drops to 2 atmospheres, which is equal to half the pressure experienced at 30 meters.
The volume doubles, right?
As we will explain in a different article, volume plays a crucial role in determining buoyancy.
When the volume of air inside the BC doubles, it results in twice the buoyant force. With such increased buoyancy, it’s only logical that the object would float up to the water’s surface.
When the pressure diminishes, it expands your range of movement, causing you to float effortlessly.
Moreover, it’s essential to exercise caution as the water becomes shallower. This will reduce pressure and increase volume.
In other words, once you lose control of buoyancy and ascent, you’ll accelerate towards the water surface.
Thus, using the BC properly gives you an important advantage. Even if you start floating up, you can quickly let out air from the BC to regain control. This helps you handle the situation and regain a stable position underwater.
(We’ll explain the risks associated with rapid ascent in a separate article).
As you move from deep water to shallower areas, the BC expands in volume, creating more buoyancy. This helps you stay afloat and maintain a comfortable position near the surface.
When moving from shallow water to deeper regions, the BC’s volume decreases, leading to a loss of buoyancy.
Hence, it becomes necessary to add air to the BC as the water depth increases.
In most license courses, about 99% of people don’t receive any specific instructions. As a result, when the guide signal “ascending~”, some divers instinctively attempt to add air to the BC.
By now, if you’ve read this far, you likely comprehend the concept, right?
During an ascent, the air within the BC expands resulting in increased buoyancy.
Thus, the appropriate action in such a situation is to “release air” from the BC!






